Introduction: myelodysplastic syndromes (MDS) are a heterogeneous group of hematological diseases. Morphologic bone marrow (BM) examination, cytogenetic analysis and molecular techniques are indispensable for accurate diagnosis and disease classification. BM constitutes the most suitable sample for molecular studies, however, peripheral blood (PB) can be obtained by minimally invasive venipuncture and would be ideal for comprehensive sequential monitoring of MDS over time. Aim: to assess whether targeted deep sequencing (TDS) allows the detection of somatic variants from diagnosis and follow-up PB samples and if the molecular profiles are comparable to those from BM samples.
Methods: preliminary results include the study of 32 low risk patients (28 MDS and 4 chronic myelomonocytic leukemia, CMML). At least one sample, either BM or PB, was obtained at the moment of diagnosis and whenever possible, paired samples obtained the same day were collected. Over the course of the disease only PB samples were obtained. Libraries were prepared for all available samples using DNA from whole BM or whole PB using a custom hybridization-probe based panel (KAPA HyperCap, Roche), including selected exons of 50 myeloid-related genes.
TDS was performed on Illumina MiSeq instrument at a mean coverage of 1000x. Data were analyzed using local bioinformatic pipeline. Only those variants with ≥100x locus coverage and ≥25 reads for the alternative allele (quality criteria) were considered for downstream analysis. Variants were filtered according to variant type, location, read depth (>100x) and population frequency (<1%).
Results: patients´ characteristics are described in table 1. The combination of cytogenetics and TDS identified genetic alterations in 29/32 patients (91%). TDS allowed the identification of somatic variants in 27/32 patients (85%), being TET2 (n=23, 29%), SF3B1 (n=12, 15%) and ASXL1 (n=8, 10%) the most frequently mutated genes.
Considering molecular information, restratification of IPSS-R to IPSS-M risk groups of MDS patients (n=28) was performed (Table 1). As previously described (Sauta et al, J Clin Oncol, 2023), we performed a five-to-five comparison of IPSS-R and IPSS-M patients' distribution (merging moderate low and moderate high to moderate in IPSS-M). This resulted in the restratification of 36% of MDS patients (10/28) of which 6% (n=2) were downstaged and 25% (n=8) were upstaged.
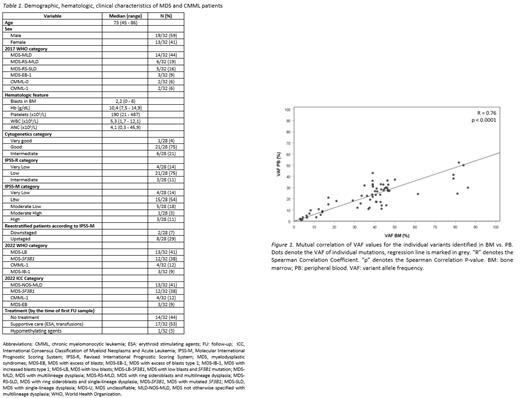
Suitability of PB samples for NGS analysis was evaluated in 21 patients in which paired BM and PB samples were obtained at diagnosis. Overall, 65 variants were found, and 63 of them were detectable in both BM and PB samples. Despite the other two variants were also detected in PB samples at low variant allele frequency (VAF) values ( TET2 and NRAS, VAF=1,5% and 0,99%, respectively), they strictly did not meet our quality criteria (≥25 reads for the alternative allele). Nevertheless, a strong correlation was identified between VAF values for the individual variants detected in BM vs. PB (Figure 1).
As part of this project, molecular profile during patients' follow-up is being studied using PB samples. To date, at least one follow-up sample of each patient was studied. The mean time between diagnosis and the first follow-up sample was 28 months (range 6-84 months). After this time, and despite molecular profiles are heterogeneous, variations could be detected in up to 72% of patients (23/32) using PB samples. The most commonly detected variation was an increasing in VAF values (9/23; defined by a difference >5% in relation to the VAF detected in the previous sample), followed by a new variant detection + increasing in VAF values (7/23), new variant detection only (6/23) and a single case where simultaneous decreasing VAF and disappearance of subclones was detected (single patient receiving treatment with hypomethylating agent).
Conclusion: molecular testing has become increasingly important for accurate diagnosis and risk assessment in MDS and CMML patients, specially with the new classifications and prognostic scoring system. Here we demonstrate that the molecular analysis from PB cells constitutes an appropriate alternative for the detection and quantification of somatic variants in MDS patients at diagnosis and during disease course.
Funding: Bristol Myers Squibb (PI-13279-IIT), FEHH and 2021 SGR 00560 (GRC).
Disclosures
No relevant conflicts of interest to declare.